SAN DIEGO, CALIFORNIA USA
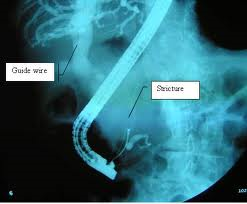
MINIMALLY
INVASIVE
Devices in the body's main cavity, treating critical organs, muscles and bones
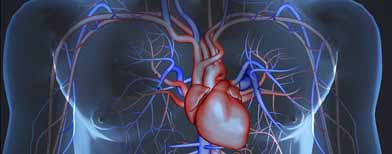
INTRAVASCULAR
Devices that treat the circulatory system, including both cardiac and peripheral vascular devices
- Cardiac catheters
- Billiary catheters
- Peripheral interventional catheters
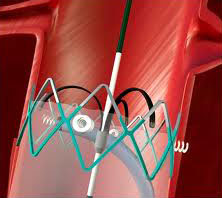
IMPLANTABLE
Devices in the body permanently, including bioresorbable implants.
- Tubing
- Strands
- Fiber
- Cast Film
directly from the low volume extrusion mixing machinery
